 Vinegar Dosing Methodology for the Marine Aquarium
Vinegar Dosing Methodology for the Marine Aquarium
Vinegar Dosing Methodology for the Marine Aquarium
Written by Cliff Babcock and Dr. Randy Holmes Farley
Why would you want to dose vinegar?
Reduce Nitrate
Proper vinegar dosing will decrease nitrate levels by increasing bacterial populations, which are then skimmed out removing the nitrate within their cells. The bacteria may also be a good food source for many organisms, including sponges, effectively taking nitrate from the water and incorporating it into the large organisms in the aquarium. The added organic matter can also be used by bacteria growing in low oxygen regions. These bacteria can use nitrate as a source of “oxygen” to metabolize the added organic, producing N2 gas. Other bacteria use the ANAMMOX process to convert nitrate to N2 gas, which dissipates out of the tank water. Vinegar promotes such processes in bacterial biofilms (masses) and other low oxygen areas. These process further reduce nitrate beyond their ordinary needs for nitrogen. This denitrification is the process that takes place inside of live rock and sand beds, and adding an appropriate organic carbon source that can penetrate these regions may spur the process.
Reduce Phosphate
Vinegar dosing like other carbon source dosing will reduce phosphate, however it is a very slow process compared to nitrate reduction. Why, because bacteria utilize omuch more nitrate than phosphate to grow and multiply. The denitrification process described above is another reason why the process may be imbalanced toward more nitrate reduction than phosphate reduction. It is the skimming out of bacteria or consumption of them by higher organisms that reduces phosphate. The ratio bacteria utilize for ordinary growth, and the ratio that likely comes in with many foods is very roughly around 16 parts of nitrate per 1 part of phosphate, which is based on the percentage of nitrogen and phosphorus present in the basic biochemicals that make up all living cells. The ratio can vary tremendously, but the point is that one should expect a lot of nitrate reduction for each unit of phosphate reduction, even if cellular growth is the only process taking place..
For these reasons, if your phosphate is high and nitrate is low, then you should use another method like GFO to reduce phosphate back in line with the ratio above. Then vinegar dosing can be used to maintain the levels of nitrate and phosphate, but is not a preferred method to reduce high phosphate levels. Many folks may need to ultimately use an additional phosphate export method, such as GFO, even if the levels start out balanced because organic carbon addition can be imbalanced to more nitrate reduction for the reasons stated above.
Enhance Coral Coloration
I have not experienced any color changes in my coral while dosing vinegar. I have not seen many posts that claim vinegar helped with color changes. Perhaps it will with some SPS coral I do not have in my tank.
Reduce Cyanobacterial Growth
Scientific research has found that cyanobacterial growth does not increase when dosing vinegar (acetate), where it was found that ethanol dosing will increase cyanobacterial growth. Cyanobacteria produce PHA to store energy when needed. PHA is an ingredient in some biopellets. So cyanobacteria can utilize some if not all of the biopellets. Hobbyists who have dosed vinegar have reported less cyanobacterial problems compared to dosing biopellets & ethanol. This was my experience as well.
Reduce Dissolved and Suspended Organics
Bacteria do utilize the dissolved and suspended organics in the water column, thus reducing the content to at least simpler forms. Bacteria will produce toxins and release them into the water column as well. Running GAC will help reduce the dissolved organics and toxins produced by bacteria.
What is the theory for nitrate & phosphate reduction using vinegar?
Bacterial growth and reproduction in a reef aquarium seem to be limited by the amount of available carbon. Simply put, vinegar adds more carbon which allows larger bacterial populations to develop when dosed. These bacteria may be wholly invisible to you when they grow hidden on rocks in refugia or on GAC, but they may also show up as blooms in the water colun or as a visible mat in the aquarium. They have to grow somewhere, so making a good place for them to grow out of sight (and possibly just upstream from a skimmer) may be a good way to prevent unsightly appearance of bacteria. When and how you dose may also impact where they grow. For example, dosing very slowly with a dosing pump just upstream from a large rock filled refugium may encourage growth there, where slow dosing the the display, or once daily bolus dosing anywhere might deliver the vinegar through the whole system and encourage visible growth in the display.
The added vinegar is encouraging bacterial growth. Therefore a good skimmer will help remove more of the free floating bacteria into the aquarium. Skimmers will not, however, remove the benthic growth of bacteria. If bacterial blooms occur it may be necessary to increase filtration with other methods such as using a diatom filter and/or filter bags.
What type of vinegar?
Use pure distilled white vinegar (5%) with no added ingredients that you buy from the grocery store. If this is not available where you live, use 98% food grade glacial acetate and dilute it down to 5%. Follow all labeled precautions when using glacial acetate since it can be hazardous.
How do you dose vinegar?
Methods for Dosing Vinegar
Vinegar does have a reducing effect on tank pH for shorter periods of time. Adding vinegar to kalk water will still have a high pH increasing effect on tank water. Therefore this is a good method to add vinegar when one is dosing kalk water to offset any reducing effect that vinegar may have. Keep in mind that when one adds vinegar to kalk water it will increase the alkalinity and calcium output when one adds more than two teaspoons (saturation point of kalk water) to 1 gallon of RODI water. For example if you add 3 teaspoons of kalk to 1 gallon of RODI water and also add 45mL of vinegar, it will increase your alkalinity and calcium output by around 1/3 - 1/2 more.
Dripping vinegar over longer periods of time is another fine method which slowly adds vinegar to tank water without increasing the pH by much. It is recommended to drip vinegar during daylight hours when pH is at its highest.
Increasing bacterial populations in tank water can rob oxygen from the tank water, so dosing the vinegar during daylight hours when oxygen is at its highest is best.
When dripping vinegar it is best to not dilute it too much or store the diluted vinegar too long since diluted vinegar can be broken down by bacteria in the dispensing vessel reducing the amount of carbon dosed. Check Coles Specials and Big W Catalogue. Dilution and delivery the same day is fine, but a 10% vinegar plus 90% water solution is not likely stable over the course of a week or two.
When vinegar is dosed in smaller amounts it is possible to add it in one or two doses per day manually. You will need to watch the pH decreasing effect to make sure it is acceptable. Again, it is best to dose in the early morning and before lights go out.
Amount of Vinegar to Dose
Dosing vinegar is similar to dosing ethanol except that vodka has more carbon in it than vinegar. The ratio is about 8 times more carbon in 80 proof (40% by weight) vodka than vinegar (which is usually 5% organic by weight). For a conservative method one may use the same guidelines for dosing ethanol in the article below, except use 8 (eight) times as much vinegar as ethanol for the 40% ethanol directions:
Vodka Dosing...Distilled!
By Nathaniel A. Walton (Genetics) and Matt Bjornson (Stony_Corals)
http://www.reefkeeping.com/issues/2008-08/nftt/index.php
Genetics subsequently modified the vodka dosing regimen for vinegar using the guidelines in link above creating the chart below. The table below can be found in a more printer friendly version here.
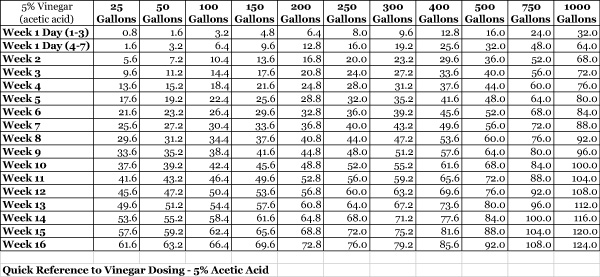 |
In my experience, I started with using 15mL vinegar per 100 gallons of water volume and had no problems. I increased weekly by 15mL vinegar per 100 gallons up to 45mL per 100 gallons. Perhaps dosing higher vinegar amounts if necessary will work as well.
Watch for bacterial blooms in the tank water. If this occurs at an unacceptable level, cut the dose back and increase filtering. Such blooms are really only a problem from the aesthetic standpoint (and possible O2 reduction). The suspended bacterial might actually be good food for many of our tank organisms, and many people seem to believe that their tank populations of sponges increase when dosing organic carbon. The acetate in vinegar can be taken up by many higher organisms, including the zoox in corals and anemones. So very high doses can lead to browning of corals and anemones through increased zoox populations. If that happens, a dose reduction is recommended.
Once nitrate and phosphate are reduced to an undetectable reading (zero) using hobby grade tests kits, I find that watching the green algae growth on tank glass is a good indicator as to when you have reached a desirable level. In my experience, I find bacterial growth replaces algal growth on the tank glass. The bacterial growth appears whitish compared to green algal growth. The bacterial growth is much easier to clean away. If bacterial growth on glass becomes a problem, reduce the amount of vinegar you dose. Keep in mind bacterial growth has to occur somewhere within your tank where it can be removed. Randy has noticed an increase in bacterial growth on his GAC between changes. Using old GAC to grow bacteria on, while dosing vinegar, would make a good mechanism for increasing bacterial growth and allow them to be exported when cleaned off using fresh salt mix.
Vinegar Dosing Articles of Interest
- What Your Grandmother Never Told You About Lime
- Expanding the Limits of Limewater: Adding Organic Carbon Sources (vinegar)
- Thread discussing some differences between carbon sources including effects on cyanobacteria
- Randy's vinegar dosing limit. I found it! (Thread)


